Defining Chirality
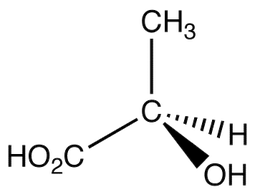
A chiral center is defined as a carbon with four different groups attached to it. As you can see to the left, no two of the substituents are the same. This means that the central carbon is a chiral carbon.
Does Lidocaine have any Chiral Centers?
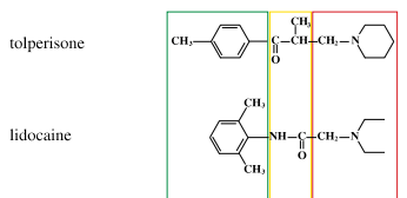
As you can see in the image to the right, we compare lidocaine to tolperisone. Notice that highlighted in the yellow box, tolperisone has a chiral carbon which has four different substituents bonded to it. Beneath that, in the same location, we see that there is no carbon within lidocaine that has four different groups attached to it. Each carbon has at least two substituents that are the same, with the exception of the C=O bond. Carbons with double bonds can not be chiral centers because they would only have three groups attached. Remember the definition of a chiral center states that a carbon must have four different groups attached. Therefore, lidocaine does not have any chiral centers.
